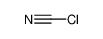
-
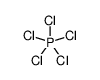
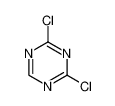
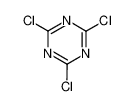
cyanuric chloride
CAS No.108-77-0
Formula:C3Cl3N3
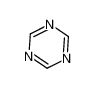
Cyanuric chloride is an organic compound with the formula (NCCl)3. This white solid is the chlorinated derivative of 1,3,5-triazine. It is the trimer of cyanogen chloride. Cyanuric chloride is the main precursor to the popular but controversial herbicide atrazine.
CYANURIC CHLORIDE FOR SYNTHESIS
Cyanuric Chloride
CYANURCHLORID
2,4,6-Trichloro-1,3,5-triazine
1,3,5-TRIAZINE,2,4,6-TRICHLORO-
CYANURIC CHLORIDE (2,4,6-TRICHLORO-1,3,5-TRIACINE)
2,4,6-TRICHLORO-S-TRIAZINE
Cyanuricchloride
2,4,6-TRICHLORO-[1,3,5]TRIAZINE
1,3,5-Triazine, 2,4,6-trichloro-
expand collapse
1.Synthesis Route
2.Synthesis Route
3.Synthesis Route
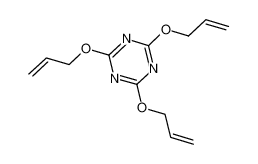
Related Compound Information
Copyright © 2015 MOLBASE All Rights Reserved.
ICP Shanghai 14014220