1.Identification
1.1 GHS Product identifier
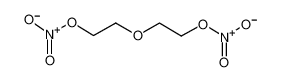
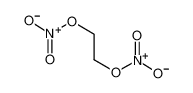
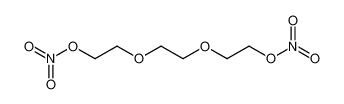
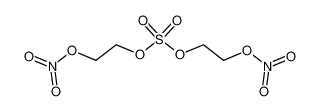
| Product name | 2-(2-nitrooxyethoxy)ethyl nitrate |
|---|
1.2 Other means of identification
| Product number | - |
|---|---|
| Other names | Oxydiethylene dinitrate |
1.3 Recommended use of the chemical and restrictions on use
| Identified uses | For industry use only. |
|---|---|
| Uses advised against | no data available |
1.4 Supplier's details
| Company | MOLBASE (Shanghai) Biotechnology Co., Ltd. |
|---|---|
| Address | Floor 4 & 5, Building 12, No. 1001 North Qinzhou Road, Xuhui District, Shanghai, China |
| Telephone | +86(21)64956998 |
| Fax | +86(21)54365166 |
1.5 Emergency phone number
| Emergency phone number | +86-400-6021-666 |
|---|---|
| Service hours | Monday to Friday, 9am-5pm (Standard time zone: UTC/GMT +8 hours). |
2.Hazard identification
2.1 Classification of the substance or mixture
no data available
2.2 GHS label elements, including precautionary statements
| Pictogram(s) | no data available |
|---|---|
| Signal word | no data available |
| Hazard statement(s) | no data available |
| Precautionary statement(s) | |
| Prevention | no data available |
| Response | no data available |
| Storage | no data available |
| Disposal | no data available |
2.3 Other hazards which do not result in classification
no data available
3.Composition/information on ingredients
3.1 Substances
| Chemical name | Common names and synonyms | CAS number | EC number | Concentration |
|---|---|---|---|---|
| 2-(2-nitrooxyethoxy)ethyl nitrate | 2-(2-nitrooxyethoxy)ethyl nitrate | 693-21-0 | none | 100% |
4.First-aid measures
4.1 Description of necessary first-aid measures
General advice
Consult a physician. Show this safety data sheet to the doctor in attendance.
If inhaled
Fresh air, rest. Refer for medical attention.
In case of skin contact
Remove contaminated clothes. Rinse and then wash skin with water and soap. Refer for medical attention .
In case of eye contact
First rinse with plenty of water for several minutes (remove contact lenses if easily possible), then refer for medical attention.
If swallowed
Rinse mouth. Give a slurry of activated charcoal in water to drink. Induce vomiting (ONLY IN CONSCIOUS PERSONS!). Refer for medical attention .
4.2 Most important symptoms/effects, acute and delayed
Excerpt from ERG Guide 112 [Explosives* - Division 1.1, 1.2, 1.3 or 1.5]: Fire may produce irritating, corrosive and/or toxic gases. (ERG, 2016)
4.3 Indication of immediate medical attention and special treatment needed, if necessary
/SRP:/ Immediate first aid: Ensure that adequate decontamination has been carried out. If patient is not breathing, start artificial respiration, preferably with a demand-valve resuscitator, bag-valve-mask device, or pocket mask, as trained. Perform CPR as necessary. Immediately flush contaminated eyes with gently flowing water. Do not induce vomiting. If vomiting occurs, lean patient forward or place on left side (head-down position, if possible) to maintain an open airway and prevent aspiration. Keep patient quiet and maintain normal body temperature. Obtain medical attention. /Ethylene glycol, glycols, and related compounds/
5.Fire-fighting measures
5.1 Extinguishing media
Suitable extinguishing media
Powder, water spray, foam, carbon dioxide. Note: Evacuate area, fight fires only from an explosion-resistant location ... Cool drums, etc., by spraying with water but avoid contact of the substance with water.
5.2 Specific hazards arising from the chemical
Excerpt from ERG Guide 112 [Explosives* - Division 1.1, 1.2, 1.3 or 1.5]: MAY EXPLODE AND THROW FRAGMENTS 1600 METERS (1 MILE) OR MORE IF FIRE REACHES CARGO. For information on "Compatibility Group" letters, refer to Glossary section. (ERG, 2016)
5.3 Special protective actions for fire-fighters
Wear self-contained breathing apparatus for firefighting if necessary.
6.Accidental release measures
6.1 Personal precautions, protective equipment and emergency procedures
Use personal protective equipment. Avoid dust formation. Avoid breathing vapours, mist or gas. Ensure adequate ventilation. Evacuate personnel to safe areas. Avoid breathing dust. For personal protection see section 8.
6.2 Environmental precautions
Evacuate danger area! Consult an expert! Collect leaking and spilled liquid in sealable containers as far as possible. Absorb remaining liquid in sand or inert absorbent. Then store and dispose of according to local regulations. Do NOT let this chemical enter the environment. Personal protection: complete protective clothing including self-contained breathing apparatus.
6.3 Methods and materials for containment and cleaning up
Evacuate danger area! Consult an expert! Collect leaking and spilled liquid in sealable containers as far as possible. Absorb remaining liquid in sand or inert absorbent and remove to safe place. Do NOT let this chemical enter the environment. (Extra personal protection: complete protective clothing including self-contained breathing apparatus.)
7.Handling and storage
7.1 Precautions for safe handling
Avoid contact with skin and eyes. Avoid formation of dust and aerosols. Avoid exposure - obtain special instructions before use.Provide appropriate exhaust ventilation at places where dust is formed. For precautions see section 2.2.
7.2 Conditions for safe storage, including any incompatibilities
Fireproof. Store in a separate building. Separated from acids and food and feedstuffs. Cool. Well closed.Fireproof. Store in separate building. Separated from acids, food and feedstuffs . Cool. Well closed.
8.Exposure controls/personal protection
8.1 Control parameters
Occupational Exposure limit values
no data available
Biological limit values
no data available
8.2 Appropriate engineering controls
Handle in accordance with good industrial hygiene and safety practice. Wash hands before breaks and at the end of workday.
8.3 Individual protection measures, such as personal protective equipment (PPE)
Eye/face protection
Safety glasses with side-shields conforming to EN166. Use equipment for eye protection tested and approved under appropriate government standards such as NIOSH (US) or EN 166(EU).
Skin protection
Wear impervious clothing. The type of protective equipment must be selected according to the concentration and amount of the dangerous substance at the specific workplace. Handle with gloves. Gloves must be inspected prior to use. Use proper glove removal technique(without touching glove's outer surface) to avoid skin contact with this product. Dispose of contaminated gloves after use in accordance with applicable laws and good laboratory practices. Wash and dry hands. The selected protective gloves have to satisfy the specifications of EU Directive 89/686/EEC and the standard EN 374 derived from it.
Respiratory protection
Wear dust mask when handling large quantities.
Thermal hazards
no data available
9.Physical and chemical properties
| Physical state | Diethylene glycol dinitrate is a liquid. Extremely sensitive explosive if not properly desensitized with a phlegmatizer. Slightly soluble in alcohol and soluble in ether. Slightly toxic by ingestion. May explode under prolonged exposure to heat or fire or from sudden shock. The primary hazard is the blast effect of an instantaneous explosion and not flying projectiles and fragments . Used as a rocket propellant. |
|---|---|
| Colour | Liquid |
| Odour | no data available |
| Melting point/ freezing point | 2(稳定体)ºC |
| Boiling point or initial boiling point and boiling range | 161°C |
| Flammability | Gives off irritating or toxic fumes (or gases) in a fire. |
| Lower and upper explosion limit / flammability limit | no data available |
| Flash point | no data available |
| Auto-ignition temperature | no data available |
| Decomposition temperature | no data available |
| pH | no data available |
| Kinematic viscosity | no data available |
| Solubility | In water, 3.9X10+3 mg/L at 25°C |
| Partition coefficient n-octanol/water (log value) | log Kow = 0.98 |
| Vapour pressure | 5.9X10-3 mm Hg at 25°C |
| Density and/or relative density | 1.38 |
| Relative vapour density | 6.76 (Air = 1) |
| Particle characteristics | no data available |
10.Stability and reactivity
10.1 Reactivity
no data available
10.2 Chemical stability
Stable under recommended storage conditions.
10.3 Possibility of hazardous reactions
A dangerous fire hazard when exposed to heat or flame.As a result of flow, agitation, etc., electrostatic charges can be generated.Nitroorganics, such as DIETHYLENE GLYCOL DINITRATE, range from slight to strong oxidizing agents. If mixed with reducing agents, including hydrides, sulfides and nitrides, they may begin a vigorous reaction that culminates in a detonation. Nitroalkanes are milder oxidizing agents, but still react violently with reducing agents at higher temperature and pressures. Nitroalkanes react with inorganic bases to form explosive salts. The presence of metal oxides increases the thermal sensitivity of nitroalkanes. Nitroalkanes with more than one nitro group are generally explosive.
10.4 Conditions to avoid
no data available
10.5 Incompatible materials
Can react vigorously with oxidizing or reducing materials.
10.6 Hazardous decomposition products
Upon decomposition it emits toxic fumes of /nitroxides/.
11.Toxicological information
Acute toxicity
- Oral: LD50 Rat male oral gavage 990 mg/kg
- Inhalation: no data available
- Dermal: no data available
Skin corrosion/irritation
no data available
Serious eye damage/irritation
no data available
Respiratory or skin sensitization
no data available
Germ cell mutagenicity
no data available
Carcinogenicity
Classification - D; not classifiable as to human carcinogenicity Basis - No human or animal carcinogenic studies found in the available literature. /Based on former classification system/
Reproductive toxicity
no data available
STOT-single exposure
no data available
STOT-repeated exposure
no data available
Aspiration hazard
no data available
12.Ecological information
12.1 Toxicity
- Toxicity to fish: no data available
- Toxicity to daphnia and other aquatic invertebrates: no data available
- Toxicity to algae: no data available
- Toxicity to microorganisms: no data available
12.2 Persistence and degradability
AEROBIC: Aliphatic nitric acid esters undergo aerobic biodegradation readily via successive removal of nitrate groups to isomeric derivatives. Nitrate esters undergo microbial metabolism by cleaving the nitrate ester group(1). An unspecified amount of diethylene glycol dinitrate was nearly completely degraded in less than 35 days when incubated in diethylene glycol dinitrate-contaminated water enhanced with the addition of yeast and glucose. Degradation in pure water was minimal, suggesting that degradation may occur in acclimated environments(SRC). Loss in combinations of sterile, nonsterile, aerobic and anaerobic river and pond sediments was all within 21 days, suggesting abiotic processes. Degradation in dry soil was slow with 16 and 24% loss after 5 weeks in sterile and nonsterile soil, respectively following the addition of 20 ppm test chemical. Using activated sludge enhanced with mineral salts, oxygen, and ethanol, diethylene glycol dinitrate was shown to degrade by hydrolytic cleavage to porducts including diethylene glycol mononitrate and diethyle glycol(2) and to finally carbon dioxide and methane(3).
12.3 Bioaccumulative potential
An estimated BCF of 3.2 was calculated in fish for diethylene glycol dinitrate(SRC), using a log Kow of 0.98(1) and a regression-derived equation(2). According to a classification scheme(3), this BCF suggests the potential for bioconcentration in aquatic organisms is low(SRC).
12.4 Mobility in soil
The Koc of diethylene glycol dinitrate is estimated as 32(SRC), using a log Kow of 0.98(1) and a regression-derived equation(2). According to a classification scheme(3), this estimated Koc value suggests that diethylene glycol dinitrate is expected to have very high mobility in soil. The sorption partition coefficients, Kp, were calculated as 2.3 and 0.8 g/mL for EPA-5 sediment (33.6% sand, 31.0% clay, and 35.4% silt at pH 7.44) and EPA-18 sediment (34.6% sand, 39.5% clay, and 25.8% silt at pH 7.76), respectively; these correspond to Koc values of 100 and 120 g/mL, respectively(1).
12.5 Other adverse effects
no data available
13.Disposal considerations
13.1 Disposal methods
Product
The material can be disposed of by removal to a licensed chemical destruction plant or by controlled incineration with flue gas scrubbing. Do not contaminate water, foodstuffs, feed or seed by storage or disposal. Do not discharge to sewer systems.
Contaminated packaging
Containers can be triply rinsed (or equivalent) and offered for recycling or reconditioning. Alternatively, the packaging can be punctured to make it unusable for other purposes and then be disposed of in a sanitary landfill. Controlled incineration with flue gas scrubbing is possible for combustible packaging materials.
14.Transport information
14.1 UN Number
| ADR/RID: no data available | IMDG: no data available | IATA: no data available |
14.2 UN Proper Shipping Name
| ADR/RID: no data available |
| IMDG: no data available |
| IATA: no data available |
14.3 Transport hazard class(es)
| ADR/RID: no data available | IMDG: no data available | IATA: no data available |
14.4 Packing group, if applicable
| ADR/RID: no data available | IMDG: no data available | IATA: no data available |
14.5 Environmental hazards
| ADR/RID: no | IMDG: no | IATA: no |
14.6 Special precautions for user
no data available
14.7 Transport in bulk according to Annex II of MARPOL 73/78 and the IBC Code
no data available
15.Regulatory information
15.1 Safety, health and environmental regulations specific for the product in question
| Chemical name | Common names and synonyms | CAS number | EC number |
|---|---|---|---|
| 2-(2-nitrooxyethoxy)ethyl nitrate | 2-(2-nitrooxyethoxy)ethyl nitrate | 693-21-0 | none |
| European Inventory of Existing Commercial Chemical Substances (EINECS) | Listed. | ||
| EC Inventory | Listed. | ||
| United States Toxic Substances Control Act (TSCA) Inventory | Listed. | ||
| China Catalog of Hazardous chemicals 2015 | Listed. | ||
| New Zealand Inventory of Chemicals (NZIoC) | Not Listed. | ||
| Philippines Inventory of Chemicals and Chemical Substances (PICCS) | Not Listed. | ||
| Vietnam National Chemical Inventory | Not Listed. | ||
| Chinese Chemical Inventory of Existing Chemical Substances (China IECSC) | Not Listed. | ||
16.Other information
Information on revision
| Creation Date | Aug 12, 2017 |
|---|---|
| Revision Date | Aug 12, 2017 |
Abbreviations and acronyms
- CAS: Chemical Abstracts Service
- ADR: European Agreement concerning the International Carriage of Dangerous Goods by Road
- RID: Regulation concerning the International Carriage of Dangerous Goods by Rail
- IMDG: International Maritime Dangerous Goods
- IATA: International Air Transportation Association
- TWA: Time Weighted Average
- STEL: Short term exposure limit
- LC50: Lethal Concentration 50%
- LD50: Lethal Dose 50%
- EC50: Effective Concentration 50%
References
- IPCS - The International Chemical Safety Cards (ICSC), website: http://www.ilo.org/dyn/icsc/showcard.home
- HSDB - Hazardous Substances Data Bank, website: https://toxnet.nlm.nih.gov/newtoxnet/hsdb.htm
- IARC - International Agency for Research on Cancer, website: http://www.iarc.fr/
- eChemPortal - The Global Portal to Information on Chemical Substances by OECD, website: http://www.echemportal.org/echemportal/index?pageID=0&request_locale=en
- CAMEO Chemicals, website: http://cameochemicals.noaa.gov/search/simple
- ChemIDplus, website: http://chem.sis.nlm.nih.gov/chemidplus/chemidlite.jsp
- ERG - Emergency Response Guidebook by U.S. Department of Transportation, website: http://www.phmsa.dot.gov/hazmat/library/erg
- Germany GESTIS-database on hazard substance, website: http://www.dguv.de/ifa/gestis/gestis-stoffdatenbank/index-2.jsp
- ECHA - European Chemicals Agency, website: https://echa.europa.eu/