
-
(S)-seviteronel
CAS No.1610537-15-9
Formula:C18H17F4N3O3
Related Compound Information
- Aminopurvalanol A,(2R)-2-[[6-[(3-Amino-5-chlorophenyl)amino]-9-(1-methylethyl)-9H-purin-2-yl]amino]-3-methyl-1-butanol
- METRIFUDIL
- Decernotinib
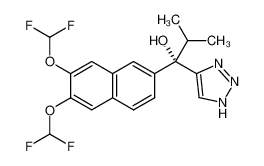
- 1-(6,7-bis(difluoromethoxy)naphthalen-2-yl)-2-methyl-1-(1H-1,2,3-triazol-4-yl)propan-1-ol
- 1-(6,7-bis(difluoromethoxy)naphthalen-2-yl)-2-methyl-1-(1H-1,2,3-triazol-4-yl)propan-1-ol
- 2-Methyl-3-(2-benzyloxyethoxy)-7-ethoxycarbonylmethylthio-pyrazolo[4,3-d]pyrimidine
- (S)-seviteronel
- Aminopurvalanol A,(2R)-2-[[6-[(3-Amino-5-chlorophenyl)amino]-9-(1-methylethyl)-9H-purin-2-yl]amino]-3-methyl-1-butanol
- METRIFUDIL
- Decernotinib

