
-
disilver,oxalate
CAS No.533-51-7
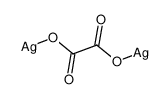
Formula:C2Ag2O4
Silver oxalate (Ag 2C 2O 4) is commonly employed in experimental petrology to add carbon dioxide (CO 2) to experiments as it will break down to silver (Ag) and the carbon dioxide under geologic conditions. It is also a precursor to the production of silver nanoparticles. It is explosive upon heating around 140 degrees Celsius, shock or friction.
Oxalic acid,disilver(1+) salt
Disilver oxalate
Silver oxalate (dry) [Forbidden]
Oxalic acid disilver salt
EINECS 208-568-3
SILVER OXALATE
Ethanedioic acid, silver(+1) salt
Silver Ethanedioate
Ethanedioic acid,disilver(1+) salt
Oxalic acid silver salt (1:2)
expand collapse
Copyright © 2015 MOLBASE All Rights Reserved.
ICP Shanghai 14014220
